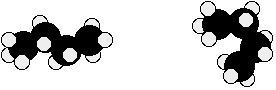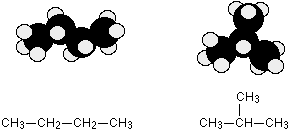|
Chemguide: Support for CIE A level Chemistry Learning outcome 13 13.3: Shapes of organic molecules; σ and π bonds Statement 13.3.1 Read the statement - it is self-explanatory. A straight chain isn't, of course, literally straight. The diagram shows butane, C4H10.
Both of the models are exactly the same molecule because the carbons and hydrogens are still joined up in the same way. But there is rotation around carbon-carbon single bonds, and the molecule can take up all sorts of different shapes in space. Even the straightest possible chain isn't actually straight, but a sort of zig-zag. And here is another form of C4H10.
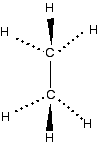
You can see that one of the carbon atoms has been taken off the end of the chain and re-attached in the middle. The right-hand model shows a branched chain. You will also come across examples of carbon atoms being joined in rings. The molecules are described as cyclic. Statements 13.3.2, 13.3.3 and 13.3.4 These statements cover the bonding and shapes of organic molecules with different types of hybridisation around the carbon atoms. Before you go on, you should find and read the statements in your copy of the syllabus. Much of this has already been covered in statement 3.4.2. Read this again before you start. Bonding and shapes of methane and ethane - sp3 hybridisation You should read the page bonding in methane which includes both methane and ethane If you are asked to draw the shape of an ethane molecule, I suggest you use something like this:
| |
|
Note: If you have forgotten what the various ways of drawing bonds mean, re-read the page on how to draw organic molecules. | |
|
The bonding and shape of ethene - sp2 hybridisation You will find everything you need on the page bonding in ethene. By the end of this, you should understand that:
The bonding and shape of ethyne - sp hybridisation Ethyne (the first member of the alkyne homologous series) isn't specifically on the syllabus, but is the simplest example of a compound with sp hybridised carbon atoms. If you followed the link to statement 3.4.2 above, you will already have read this, but refresh your memory by looking again at the formation of sp hybrid orbitals by reading the page about bonding in ethyne. Predicting shapes of similar molecules As a general guide, remember that:
© Jim Clark 2020 |
|