|
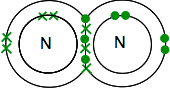
Chemguide: Support for CIE A level Chemistry Learning outcome 3.4.1 Covalent bonding and coordinate (dative covalent) bonding This statement deals with covalent bonding and coordinate (also known as dative covalent) bonding. Learning outcome 3.4.1(a) This part of the statement wants you to look at covalent bonding in a number of simple molecules. I suggest you make a list of these and tick them off when you have read about them. The Chemguide pages that you will read include almost all of these, but not in the order in the syllabus. You will find the ones that aren't included after the links. Start by making a note of the definition of a covalent bond from the syllabus. You should read a part of the page which looks at covalent bonding - single bonds. Stop when you get to the heading which says "A more sophisticated view of covalent bonding". We will come back to that later on in this section. Up to that point, you will have come across hydrogen, chlorine, hydrogen chloride, methane and ammonia from the syllabus statement. Then follow the link at the bottom of the page to find out about double bonding in oxygen, carbon dioxide and ethene. All you need to read for now on that page is the first section. For the time being, you can ignore everything when it starts to get more sophisticated. Extras to those pages This is mentioned on the second page, but not on the first. A pair of shared electrons is shown by a single line drawn between two atoms. It only applies to covalent bonds. A double bond is shown by two lines, and a triple bond by three lines. You should never draw a line between two ions - you would be penalised in an exam for doing that. The two molecules not covered on those two pages are nitrogen, N2, and ethane C2H6. Nitrogen has a triple bond between the atoms, the nitrogens sharing three pairs of electrons.
This is much easier to see using lines to represent pairs of shared electrons:
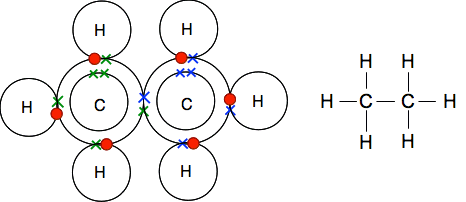
Ethane contains only single bonds, and is just slightly more complicated than, say, methane.
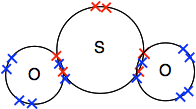
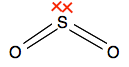
Learning outcome 3.4.1(b) This statement points out that some elements can have more than 8 electrons in their outer level when they form covalent bonds. The elements are said to have "expanded their octet". There is no suggestion in the syllabus that you should be able to explain this. The compounds asked for in the syllabus are: Sulphur dioxide The dot-and-cross diagram (showing only the outer electrons) looks like this:
This is quite awkward to draw tidily, and is better represented by a drawing with lines showing the pairs of shared electrons - but if you do that, you need to include the lone pair at the bonding level.
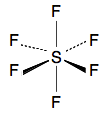
If you count the electrons around the sulfur at the bonding level, you will find there are 10, not the more normal 8. Phosphorus pentachloride You will have seen a dot-and-cross diagram for this on the page you will read previously. Sulfur hexafluoride I am not even going to attempt a dot-and-cross diagram for this! In the line diagram below, there are some conventions that you probably won't have met before. A simple straight line shows a bond in the plane of the paper or screen. A dotted line shows a bond going back behind the paper or screen. A wedge shape shows a bond coming out towards you. This helps you to make sense of the shape of a 3D molecule in 2D.
Each line (of whatever type) represents 2 electrons. In this molecule, the sulfur has expanded its octet to 12 electrons. Learning outcome 3.4.1(c) This covers coordinate covalent bonding, also called dative covalent bonding. It doesn't matter which of these terms you have been taught - you must be familiar with both of them, and realise that they mean exactly the same thing. | |
|
Note: You will find two versions of the word coordinate, alternatively written as co-ordinate. The syllabus uses "coordinate". I tend to use the version with the hyphen, although I'm not entirely consistent - you may find both versions on Chemguide. Either is OK. | |
|
You should read a part of the page which looks at co-ordinate (dative covalent) bonding. The syllabus only mentions the ammonium ion, and the bonding in the aluminium chloride dimer. That means that you must be familiar with both of those. However, you might be asked about other things as well if they involve the same principle of one atom donating both electrons to the bonding pair. So look at the other examples, and make sure they make sense to you. Make sure that you understand everything down as far as (but not including) "The bonding in hydrated metal ions". You will eventually have to know about hydrated metal ions, but not yet.
© Jim Clark 2019 |
|