|
Chemguide: Support for CIE A level Chemistry Learning outcome 3.4.3 An introduction to bond energy and bond length This statement Is a very brief introduction to bond energy and bond length in covalent molecules. Learning outcome 3.4.3(a) This statement just wants definitions. Bond energy is confusingly known by a number of different names. It is also known as bond enthalpy, bond dissociation enthalpy and bond strength. Whichever name it goes by, it means exactly the same thing.
In other words, the energy required to do break one mole of bonds in this process:
The point about everything being in the gas state is essential. When you come to do calculations with it in a future section, you cannot use bond energies to do calculations directly from substances starting in the liquid or solid state. As an example of bond energy, to break up 1 mole of gaseous hydrogen chloride molecules into separate gaseous hydrogen and chlorine atoms takes 432 kJ. The bond energy for the H-Cl bond is +432 kJ mol-1. The plus sign shows that energy is needed to make this change. Bond length This is just obvious.
Learning outcome 3.4.3(b) This statement looks at how bond energy and bond length can affect reactivity. A good example of this comes from a group of organic compounds that you will meet in detail later in the course. These compounds have a halogen atom (fluorine, chlorine, bromine or iodine) attached to a hydrocarbon chain. These compounds are known as halogenoalkanes (or previously as alkyl halides). A simple set of these with varying halogens would be
An important part of their chemistry is the replacement of the halogen atom with some other group of atoms. To do that needs you to break carbon-halogen bonds. Bond length As the halogen atom gets bigger the bond length increases.
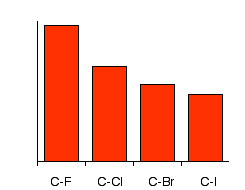
Is this surprising? Of course not! The halogen atoms are getting bigger as you go down the list - there are more layers of electrons. The distance between the nuclei is bound to increase. The importance of bond energies The pattern in bond energies of the four carbon-halogen bonds is:
Notice that bond energy (the energy needed to break the bond) falls as you go from C-F to C-I, and notice how much stronger the carbon-fluorine bond is than the rest. | |||||||||||
|
Note: You will find almost as many different values for bond strengths (or bond enthalpies or bond energies) as there are different sources! Don't worry about this - the pattern is always the same. This is why you have got a chart here rather than actual numbers. | |||||||||||
|
So, why do the bonds get weaker as you go down this series? A covalent bond is formed by the nuclei of both atoms involved being attracted to the same shared pair of electrons. If the bond is getting weaker, then that attraction must be weakening. That is simply because as the bond gets longer, the shared electron pair is getting further from one of the nuclei because of the extra layers of inner electrons being added. You might argue that the halogen nuclei are getting more positively charged because of the additional protons. But each increase in number of protons is exactly offset by the same number of inner electrons between the nucleus and the bonding pair. That extra shielding cancels out the effect of the extra protons. The bonds are getting weaker because they are getting longer. What about the relationship with reactivity? In order for anything to react with the halogenoalkanes, the carbon-halogen bond has got to be broken. Because that gets easier as you go from fluoride to chloride to bromide to iodide, the compounds get more reactive in that order. Iodoalkanes are the most reactive and fluoroalkanes are the least. In fact, fluoroalkanes are so unreactive that they are normally ignored when you talk about this set of compounds.
© Jim Clark 2019 |
|||||||||||