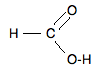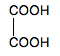|
Chemguide: Support for CIE A level Chemistry Learning outcome 33: Carboxylic acids and derivatives 33.1: Carboxylic acids Learning outcome 33.1.3 This statement is about the oxidation of methanoic and ethanedioic (oxalic) acids. Before you go on, you should find and read the statement in your copy of the syllabus. Statement 33.1.3(a): Oxidising methanoic acid Methanoic acid, HCOOH, has this structure:
If you look at the top half of this, you will see an aldehyde group, with a hydrogen attached to a carbon-oxygen double bond. Aldehyde groups can be oxidised using things like Fehling's solution or Tollens' reagent. The reactions of methanoic acid look just the same as those with aldehydes. You will find these on the page oxidation of aldehydes and ketones. The methanoic acid is oxidised to carbon dioxide and water. With Fehling's solution you get a red precipitate of copper(I) oxide
and with Tollens' reagent you get a silver mirror.
With acidified potassium dichromate(VI) solution, the orange solution would change to green. With acidified potassium manganate(VII) solution, the purple solution would become colourless.
Notice that in every case, the methanoic acid is oxidised to carbon dioxide and water. This was new for the 2016 syllabus, and I could only find a single question on it in all the papers from 2016 to the end of 2019. That just expected you to know that methanoic acid was oxidisable by these reagents and quote a reaction and colour change of your choice. Just two marks! You are probably unlikely to need the more detailed equations in any of these. Statement 33.1.3(b): Oxidising ethanedioic (oxalic) acid Ethanedioic acid is:
It is oxidised to carbon dioxide by warm potassium manganate(VII) solution acidified with dilute sulfuric acid. The reaction is often used to standardise potassium manganate(VII) solution. You can't make up a solution of potassium manganate(VII) with an exactly accurate concentration, but you can with ethanedioic acid. A standard solution of ethanedioic acid is acidified with dilute sulfuric acid, warmed and titrated with potassium manganate(VII) solution from a burette. The potassium manganate(VII) solution is decolourised until you get to the end point when a trace of pale pink remains. You can then do a calculation to find the actual concentration of the potassium manganate(VII) solution. It is possible that you might need to write an equation for this in a practical context, and you will have to build it up from half-equations. The half-equation for the ethanedioic acid is
The half-equation for the manganate(VII) ion is
If you combine those it gives:
Check that you can do this! If you can't get it right, you need to spend some time looking at the page about writing equations for redox reactions. This example is similar to the hydrogen peroxide and manganate(VII) ion case.
© Jim Clark 2020 |