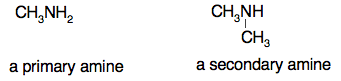|
Chemguide: Support for CIE A level Chemistry Learning outcome 33: Carboxylic acids and derivatives 33.3: Acyl chlorides Learning outcome 33.3.2 This statement is about the reactions of acyl chlorides and is in several parts. I suggest you read the individual parts of the syllabus as you go along. Important background work Before you start it is important that you understand what acyl chlorides are before you look at detailed chemistry. Read the page introducing acyl chlorides. Learning outcome 33.3.2(a) This refers to the reaction of acyl chlorides with water. You will find this, and much more, on the page about acyl chlorides and water, alcohols or phenol. Read the introduction, but the important bit is the short section about the reaction with water. You will need the rest of this page for later parts of this statement. Learning outcome 33.3.2(b) Now continue on the same page to read about the reactions with alcohols. You will also find this discussed on the page about the preparation of esters starting about half-way down the page. Concentrate on the reactions using acyl chlorides. Ignore entirely the section about acid anhydrides. Learning outcome 33.3.2(c) Again continue on the page about acyl chlorides and water, alcohols or phenol to read the section on the reaction with phenols. Although the syllabus talks about "phenol", it would be typical of CIE to ask you a question which involved a more complicated example such as the aspirin one at the bottom of the page. I am not suggesting that it is important for you to learn this example, but it is essential that you understand it. Learning outcome 33.3.2(d) This statement is about the reaction between acyl chlorides and ammonia. You will find this on the page about the reaction of acyl chlorides with ammonia or primary amines. Just read down as far as the ammonia reaction for now. Warning! The syllabus specifies the formation of an amide and HCl. It does not expect you to add the follow-up reaction with excess ammonia. Mark schemes from past papers follow the same line. This applies to the next statement as well.Learning outcome 33.3.2(e) This statement extends the previous one to the reaction of acyl chlorides with primary and secondary amines. Primary amines are essentially ammonia molecules in which one of the hydrogens has been replaced with a hydrocarbon group. A secondary amine has two of the hydrogens in an ammonia molecule replaced by a hydrocarbon group. For example:
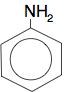
Phenylamine, an aromatic amine, is also a primary amine. The hydrocarbon group attached to the nitrogen doesn't have to be an alkyl group.
So, continue reading the page about the reaction of acyl chlorides with ammonia or primary amines to the end of the page. The same warning applies as with the ammonia case. CIE just want you to produce the N-substituted amide and HCl. In the phenylamine case, that would be:
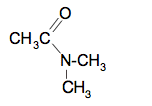
The syllabus also talks about secondary amines which aren't covered on that Chemguide page. The equation for the dimethylamine (whose formula is above) is:
The structure of the organic product is:
In every case, a hydrogen attached to the nitrogen and the chlorine in the COCl group come off as HCl, and what's left over joins together.
© Jim Clark 2020 |