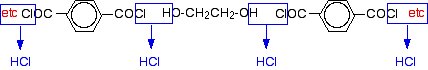|
Chemguide: Support for CIE A level Chemistry Learning outcome 35: Polymerisation 35.1: Condensation polymerisation Learning outcome 35.1.1 This statement is about the reactions used to make polyesters - a type of condensation polymerisation. Before you go on, you should find and read the statement in your copy of the syllabus. General Condensation polymerisation involves monomers joining together with the loss of a small molecule such as water or HCl. There are two common cases where this happens. One involves the formation of ester linkages to give polyesters such as Terylene; the other involves the formation of amide linkages to give polyamides such as nylon. Proteins and other polypeptides are also formed by condensation polymerisation of amino acids. Statement 35.1.1(a) This statement is about the formation of polyesters like Terylene. Read the beginning of the page about polyesters. You do not need to know about their manufacture. You also don't need to know about the hydrolysis of polyesters for this statement, but will need it for a later one. Work out the structure of the polyester by putting it together from the monomers as shown in the diagram with the water molecules being removed. You should also be aware that you could make polyesters using a molecule containing two -COCl (acyl chloride) groups as one of the monomers. The difference this time would be that you would lose HCl rather than water. For example, you could make Terylene by:
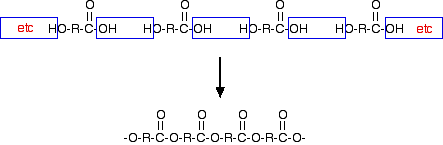
Statement 35.1.1(b) Yet another way of making a polyester (but not Terylene this time) would be to use a single monomer with a -COOH group at one end and an -OH group at the other. For example:
"R" could be any hydrocarbon group - a chain or a ring.
© Jim Clark 2020 |