|
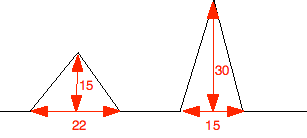
Chemguide: Support for CIE A level Chemistry Learning outcome 37: Analytical techniques 37.2 Gas / liquid chromatography Learning outcomes 37.2.1, 37.2.2 and 37.2.3 These statements are about gas-liquid chromatography. Read the individual statements as we go through them. To start with, read the whole page about gas-liquid chromatography so that you understand the background to gas-liquid chromatography. You need to understand this in principle, but do not need any of the practical detail. You particularly need to make sure that you understand the importance of retention time, because that is specifically mentioned twice in the syllabus. You must be aware of the reasons that substances will have different retention times. Statement 37.2.1 Read the statement. It tells you all you need to know. Again, make sure that you understand the term "retention time". Notice also that the syllabus specifically mentions the stationary phase consisting of a non-polar liquid. In fact, stationary phases of differing degrees of polarity are also available for analysing different types of mixtures. But if CIE want a non-polar stationary phase, that is what you give them. Statement 37.2.2 This asks you to work out percentage composition of a mixture from a gas-liquid chromatogram. In the best case, they will give you a table of areas under the curve, and ask you to find the percentage of one of the components. In the worst case, you could get a simplified diagram like this, and you would need to measure it on the exam paper. The diagram shows the sort of results that you might get. Measure it as accurately as you can.
The numbers are in mm. (The diagram is only a sketch. Please don't tell me if you measure them and find they are something slightly different!) What you need to do is to find the areas under the two peaks, add them together, and then work out the percentages. To do that, you need to remember a simple bit of geometry - the formula for the area of a triangle. That's 1/2 x base x height. So in this case, the two areas are
That gives a total area of 390 mm2. From this, you can work out the percentage of the total area due to each component.
Don't expect more than 1 mark for doing this! Statement 37.2.3 This is again about retention time, and wants you to explain why retention times differ. The syllabus talks about ". . . in terms of interactions with the stationary phase". This is covered in the page you have read, and will mainly be because of the solubility of a particular component in the stationary phase. A low solubility will mean it spends more time in the mobile gas phase, and so the retention time will be shorter than for a more soluble component which is spending more time dissolved in the stationary phase. Although it isn't actually mentioned in the syllabus, you should also be aware of the effect of the temperature of the column. That is also covered in the page you have just read.
© Jim Clark 2020 |