|
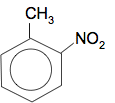
Chemguide: Support for CIE A level Chemistry Learning outcome 37: Analytical techniques 37.3 Carbon-13 NMR spectroscopy Learning outcomes 37.3.1 and 37.3.2 Start by reading the page What is C-13 NMR?. From that page, the essential thing is to realise that each line in a C-13 NMR represents a carbon atom in a different environment. You will also need to know about TMS and the solvent used. You can ignore completely the long green box at the bottom of the page. Now read the page Interpreting C-13 NMR spectra. It is probably safe to say that if you can work out the number of lines a given structure would produce, and use a table of chemical shifts, that is probably all you will need. Don't worry too much about the reasons for the different chemical shifts. If you need it, you will be given a table of chemical shifts in an exam. You will find a copy of the table towards the bottom of the syllabus. As you read through this NMR page, refer to that table rather than the one which I give. You need to get used to the table that CIE uses. The most common CIE questions have given you a structure (probably unfamiliar) and asked you to predict the number of peaks in its C-13 NMR spectrum. That sounds easy, but can actually be quite confusing - so we will talk through a few examples. Example 1 2-nitromethylbenzene has the structure:
How many lines would there be in the C-13 NMR spectrum? You need to work out how many different environments the carbon atoms are in. The carbon in the CH3 group is obviously in a different environment from all the others - and so is the one in the ring which is attached to that CH3 group. The carbon in the ring attached to the NO2 group is also obviously in a different environment from all the other carbons. What about the other carbon atoms in the ring? This is where you have to be very careful! When you are working this out, you have to worry not just about the immediate neighbour of any carbon atom, but what happens further around the ring. If you trace around the ring from the other four carbon atoms, you get a different pattern in each direction from all of them. These four carbon atoms are all in slightly different environments. If you count all this up, there are 7 carbon atoms in 7 different environments, and so 7 lines in the C-13 NMR spectrum. Example 2 4-nitromethylbenzene has the structure:
How many lines would there be in the C-13 NMR spectrum? Superficially, this looks the same as the previous example, but it isn't. It is the same for the CH3 group and the carbon it is attached to, and it is the same for the carbon attached to the NO2 group. But it isn't the same for the other carbons in the ring. The carbon atoms in the 2- and 6-positions are in exactly the same environment. If you trace around the ring from each in both directions, they are joined up in identical ways. These two carbons would produce exactly the same line. Similarly the carbons in the 3- and 5-positions are in exactly the same environment, and again would only produce a single line. That means that there will be a total of 5 lines in the C-13 NMR spectrum. Something which helps is to look carefully at the various carbon atoms in a compound for the presence of symmetry. 4-nitromethylbenzene does have some symmetry (the left-hand side is a reflection of the right-hand side); 2-nitromethylbenzene doesn't. | |
|
Note: If you actually find these two spectra on the SDBS database (mentioned in the pages you have looked at), you will find that some of the lines are so close together that they look like a single line. You need to refer to the table of line data underneath for an accurate picture. | |
|
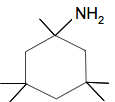
Example 3 This comes from a CIE past paper - March 2019 paper 42 Q6(c)(i). The molecule in question was:
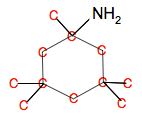
If you aren't totally happy with skeletal formulae, remember that there is a carbon atom at every junction of lines and at the end of any line with nothing else already written there. You might find it helpful to write in the carbon atoms:
By being drawn the way it is, the diagram is potentially misleading. If you look at the top of the diagram, it seems as if there is no symmetry in the molecule. There seems to be a CH3 pointing in one direction and an NH2 in the opposite direction. That isn't true! The carbon atom in the top of the ring has a tetrahedral arrangement of other atoms around it. That means that those two groups would be pointing one into and one out of the paper or screen. Ideally the bonds to the two groups would be shown as a wedge and a dotted line.
This is actually also true of the 4 CH3 groups attached lower down the ring, but it doesn't matter so much with them - they look symmetrical in the diagram anyway. So there is symmetry in the structure if you sliced vertically down through the molecule. So, let's count up the number of carbon atoms in different environments: The two carbons at the very top of the structure are in unique positions - the top carbon in the ring, and the carbon in that CH3 group. The two carbons in the 2- and 6-positions in the ring are both in the same environment. If you trace around the ring in both cases, you meet exactly the same pattern of other atoms or groups. So there is only one line for those two carbons. That is also true of the next carbon atoms in the ring - the ones with the CH3 groups attached. They are in exactly the same environment as each other, and so again only produce one line. The bottom carbon in the ring is in a unique position and so produces one line. The four carbons in the CH3 groups are all in an identical environment and so only produce a single line. Adding all this up you have a total of 6 lines in the spectrum. None of the examples above, and all the other equivalent ones you might find, are worth more than 1 mark, and so you must be able to come up with a correct answer in about 1 minute. The only way you are going to be able to do this is to practise with all the questions you can find. C-13 NMR first appeared in CIE exams in the 2016 sessions, but the majority of papers since then have had an example - mostly of the sort we have just been looking at. It is possible that you might meet questions in which you have to use data about the δ shift. In the past this has been provided in a Data Booklet given to you in the exam. In this syllabus, it looks as if this data will be included in the question paper judging by the specimen paper.
© Jim Clark 2020 |
|