|
Chemguide: Support for CIE A level Chemistry Learning outcome 2.3.1 Formulae This statement deals with writing formulae for ionic compounds. Before you go on, you should find and read the statement in your copy of the syllabus. Working out formulae for ionic compounds is something that you should have met and been comfortable with in whatever course you did between the ages of 14 and 16, and you may feel happy to skip through most of this section. But don't be over-confident! How writing formulae for ionic compounds works First of all, of course, you need to know that the compound is ionic. Secondly, all these ionic compounds are overall electrically neutral. There have to be equal numbers of positive and negative charges in the compound. How do you know if a compound is ionic? A compound will be ionic if . . .
How do you know if an ion is positive or negative?
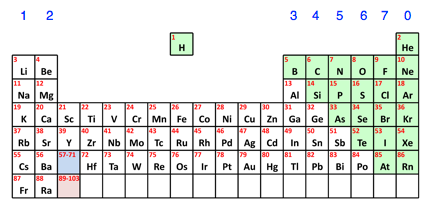
How do you know if something is a metal or non-metal? There is a simple pattern from the Periodic Table. Here is a simplified version of the Table missing out the bits that aren't relevant at the moment. The non-metals are shown in green.
You can see that the non-metals are all found on the right-hand side of the Periodic Table. All the rest of the elements are metals. | |||||||||||||
|
Note: The dividing line between metals and non-metals isn't quite as clear-cut as this. Some of the elements just to the left of the green bit tend to have properties which are a mixture of metal and non-metal. This isn't something you need to worry about at the moment. | |||||||||||||
|
How do you name non-metal ions? Simple ions from non-metals have their endings changed to "ide". So the ions are called nitride, oxide, sulfide, fluoride, chloride, bromide, and iodide. Not all of the non-metals form simple ions. The ones mentioned above are the only simple ions you are likely to come across. Phosphorus does form a phosphide ion, but it isn't common. | |||||||||||||
|
Note: Are there non-simple ions? Yes! "Complex ions" are also common in the non-metals. These contain the non-metal plus other things as well - often oxygen. We will have more to say about this below. | |||||||||||||
|
How do you know how many charges a positive ion has? Where the name of the compound tells you the number of charges on a metal ion Quite a lot of compound names have a Roman numeral as a part of the name.This is known as the oxidation state or oxidation number of the element. For example . . .
This tells you how many positive charges the ion has. We are talking about the charges on a metal ion, and so they will always be positive. So . . .
Where you can work out the number of charges on a metal ion from a Periodic Table Let's look again at the Periodic Table from earlier on.
The numbers at the top of the Groups (the vertical columns in the table) are the old group numbers - from 1 to 7 and then 0. These ignore the transition metals. This has the big advantage for students at this level that it counts the number of electrons in the outer energy level of the atoms of each element. | |||||||||||||
|
Note: The last Group in the Periodic Table (the Noble Gases) is usually called Group 0, but was sometimes called Group 8. Helium, of course, only has room in its outer level for 2 electrons. | |||||||||||||
|
Modern numbering includes the transition metals, and the numbers go from 1 to 18. CIE use this modern system, but it has a disadvantage when using the Periodic Table to work out charges on ions. To convert the new numbering to the old one, just subtract 10 from groups 13 to 17. Why does this matter? For the metals in Groups,1 2 and 3 (on the old numbering) the number of charges on the ions is the same as the Group number. That is because they have 1, 2 or 3 electrons in their outer levels to give away to something else. So. . .
The positive ions you will need to learn Group 1, 2 and 3 metals are easy, and so are all those with the Roman numerals in their name. There are a couple of metals, though, where the Roman numerals are left out more often than not. You will need to learn . . .
Don't forget that you will also need to know about these positive ions which don't contain metals . . .
Working out the charges on non-metal ions using the Periodic Table We are talking here about simple non-metal ions. The ones you are most likely to come across are oxide, sulfide, fluoride, chloride, bromide, and iodide in Groups 6 and 7. Elements at the top of Group 5 (nitrogen and phosphorus) also form negative ions in rare cases - so you should know how to work out the charges on nitride and phosphide ions as well just in case.
Elements in Groups 5, 6 and 7 have 5, 6 and 7 electrons in their outer energy levels. That means they have room to gain 3, 2 or 1 electrons to form ions with 8 electrons in the outer level.
| |||||||||||||
|
Note: For Group 5, this only really applies to nitrogen and to an even smaller extent phosphorus. Most of time, nitrogen and phosphorus form covalent bonds, or are a part of a complex ion (see below for one with nitrogen in). | |||||||||||||
|
Complex negative ions A complex ion is one made up of more than one sort of atom.
You just have to learn these. Some worked examples You need to have a copy of the Periodic Table available which has the names of the elements as well as their symbols. As you read through these examples, work them out for yourself before you read on. The more you pactise this, the easier it will become. What is the formula for chromium(III) chloride? The name tells you that the chromium has 3+ charges. The ions are therefore Cr3+ and Cl-. To balance the charges on the chromium you will need 3 chloride ions. The formula is CrCl3. What is the formula for potassium sulfate? Potassium is in Group 1 and so forms a 1+ ion. The ions are therefore K+ and SO42-. To balance the charges on the sulfate ion you will need 2 potassium ions. The formula is K2SO4. What is the formula for iron(III) oxide? The name tells you that the iron has 3+ charges. The ions are therefore Fe3+ and O2-. This is slightly more tricky! The only way to balance the charges is to have 2 iron(III) ions and 3 oxide ions: 6 pluses and 6 negatives. The formula is Fe2O3. What is the formula for magnesium nitrate? Notice this is magnesium nitrate, NOT magnesium nitride - the ends of the words really matter. The nitrate ion is NO3-. Magnesium is in Group 2 and so has 2+ charges, Mg2+. To balance the charges, you will need two nitrate ions for every magnesium ion. The formula is Mg(NO3)2. This is important! Notice the brackets around the nitrate group. If you need to have more than one complex ion, you must enclose it in brackets before you write any number after it. The "2" in the magnesium nitrate formula has to apply to the whole of the nitrate group. The only way of showing that is to enclose it in brackets. However, if you only need one of the group, you must not put brackets around it - that would be wrong. See, for example, the formula for potassium sulfate above. There are no brackets in this, because there is only one sulfate group. It is essential that you can write formulae for ionic compounds reliably. You can make yourself look really silly in an exam if you get simple formulae wrong.
© Jim Clark 2019 |
|||||||||||||